Which is correct for an adiabatic process?Īdiabatic machine should be completely insulated from the surroundings. Where A is the cross-sectional area of the piston and dV Adx is the increase in the volume. Is an adiabatic process always reversible?Ī reversible, adiabatic process is always isentropic since no entropy generation because of irreversibilities (sgen=0) and no change of entropy due to heat transfer (ds=? Q/T=0). Therefore, the change in the entropy for an adiabatic process equals to zero. The pressure and volume at point A is PA. Patm The pressure and volume at point D is PD 101 kPa and Vp 5.00 m³. O Question: Question 3 In the PV diagram below, we have a quasi-static adiabatic process going from point D to A. what is the work done on the gas going from point D to A O-326 kJ.

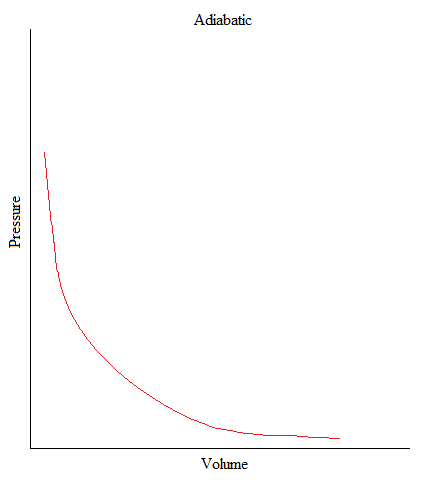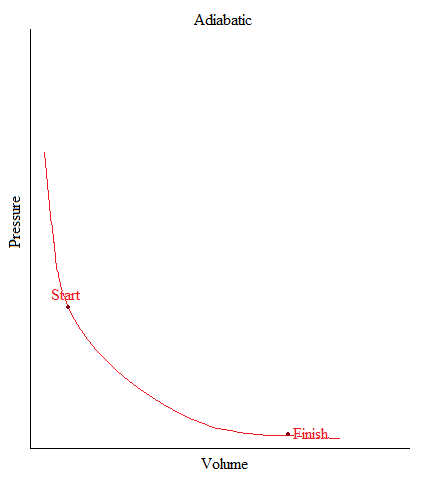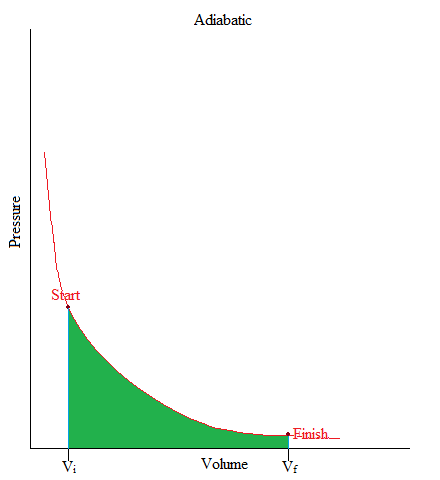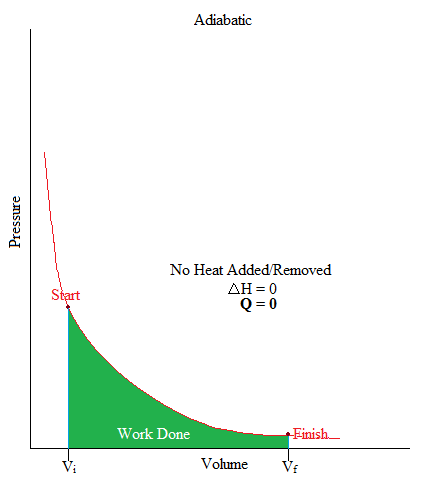
In the worked solutions the work equation seems to be reversed for the answer, as can be seen in the screenshot- the top row is p1v1-p2v2 insted of p2v2-p1v1. Thus The trade in entropy between the S1 and S2 could be equivalent to zero. If this is an ideal diatomic gas (y 1.4). I am completing a question that is requiring calculation of the work done in a polytropic process. Is Delta's zero for an adiabatic process?Īs no warmth is authorized to switch between the encompassing and machine, the warmth stays constant. Calculate the general temperature, the paintings achieved, and the alternate in interior power when 0.0400 moles of CO at 25.0 oC undergoes a reversible adiabatic enlargement from 200. Solution:- First we have to find out final pressure pf. Finally, the heat and the internal energy change over the entire cycle are respectively: Since the cycle goes clockwise, the work done by the gas is. Furthermore, since it is a compression, the work done by the gas is negative. What is ∆ U in adiabatic process?Īccording to the definition of an adiabatic process, ΔU=wad. In an adiabatic process, the final pressure pf is defined as, pf piVi/Vf pi Vi/Vf The work done W for adiabatic process is defined as, W pfVf - piVi/ (-1) Here, initial pressure is pi, initial volume is Vi and final volume is Vf and adiabatic constant is. Adiabatic process 3-1: During this adiabatic compression, the internal energy increases since the temperature rises. Let W be the work done when the system goes from the initial state (P i, V i, T i) to the final state (P f, V f,T f) adiabatically. A frictionless and insulating piston of cross sectional area A is fitted in the cylinder. Adiabatic Process - Work, Heat & Internal Energy, Gamma Ratio. Work done in an adiabatic process: Consider µ moles of an ideal gas enclosed in a cylinder having perfectly non conducting walls and base. Thus for a reversible adiabatic process and a perfect fuel, C VdT = −PdV. Derivation of expression for work done in Adiabatic process. An adiabatic process is one through which no heat enters or leaves the machine, and hence, for a reversible adiabatic process the primary law takes the shape dU = − PdV.
